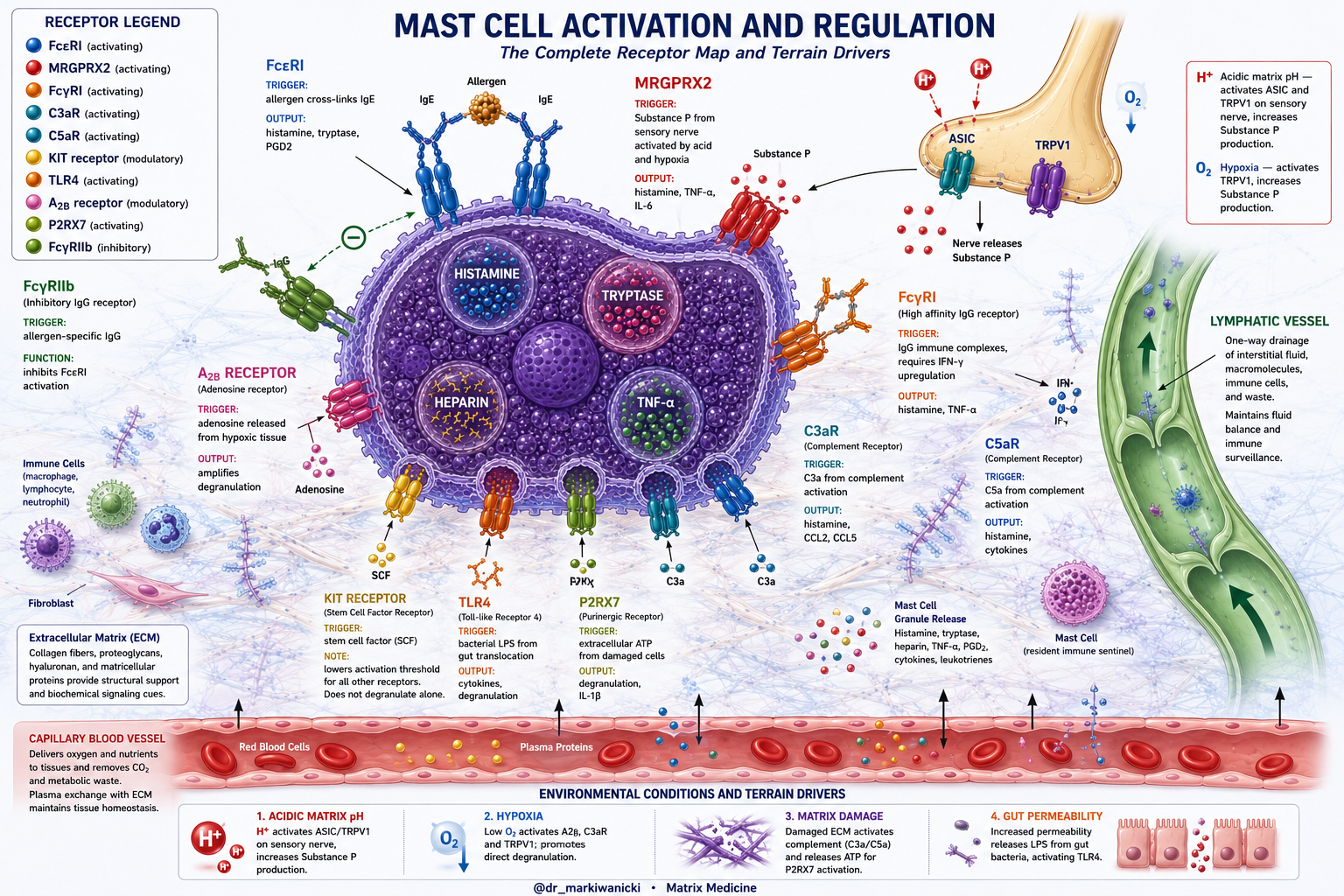
Most practitioners learned one pathway in mast cell biology. The IgE pathway. An allergen cross-links IgE antibodies on the FcεRI receptor, calcium floods the cell, and granules release histamine, tryptase, and prostaglandins.
But in patients with mast cell activation syndrome, chronic urticaria, histamine intolerance, or complex inflammatory conditions, the IgE pathway rarely tells the complete story. Mast cells express multiple activation receptors, each responding to a different upstream signal, and in chronically ill patients several of these pathways are running simultaneously.
Understanding the complete receptor map, and the soil those mast cells are living in, is one of the most clinically important shifts a practitioner working with complex chronic patients can make.
The Two Main Activation Pathways
Pathway 1: FcεRI, The IgE Pathway
The FcεRI receptor is the high affinity IgE receptor and the primary pathway taught in conventional training. Allergen cross-links IgE on the receptor surface, calcium floods the cell, and preformed granules release their contents within seconds. This is specific, episodic, and allergen dependent. Antihistamines, cromolyn sodium, and ketotifen work primarily through this pathway.
Pathway 2: MRGPRX2, The Terrain Pathway
MRGPRX2 requires no allergen and no IgE. It fires in response to Substance P released from sensory nerve fibers embedded directly in the matrix alongside the mast cells they activate. Research confirms that MRGPRX2-positive mast cells actively migrate toward and reside adjacent to Substance P-positive nerve fibers, forming a functional nerve-mast cell unit within the matrix.
What activates the nerve is the quality of the matrix. Acid-sensing ion channels ASIC and TRPV1 on sensory nerve terminals fire when local pH drops, exactly what happens in a congested matrix accumulating metabolic waste. The nerve releases Substance P. Substance P binds MRGPRX2. The mast cell degranulates without any allergen involved.
In a chronically acidic, hypoxic, poorly drained matrix the nerve-mast cell unit never fully deactivates. Substance P remains chronically elevated. The mast cell fires in response to the terrain itself. Heat, cold, food, stress, fragrance, any input in an already maximally sensitized environment crosses the threshold.
This is why MCAS patients react to everything. The terrain is the trigger.
The Additional Activation Pathways
Beyond FcεRI and MRGPRX2, mast cells express multiple additional receptors all directly relevant to chronic inflammatory presentations:
FcγRI: The IgG Immune Complex Pathway Upregulated by IFN-γ in chronic inflammatory conditions. IgG immune complexes formed against food proteins, environmental antigens, or microbial components cluster this receptor and trigger degranulation independent of IgE. This mechanistically explains why food sensitivity patients with elevated IgG reactions experience mast cell symptoms despite negative allergy testing.
C3aR and C5aR: The Complement Pathways The complement system generates C3a and C5a anaphylatoxins continuously in damaged, congested matrix tissue through the alternative pathway. C3a is one of the most potent mast cell activators identified in human studies. A congested matrix is a continuous low-grade complement activation environment. C3a also recruits additional mast cells to the site, concentrating more reactive cells in the most degraded terrain.
KIT Receptor: The Threshold Lowerer Stem cell factor binding to KIT does not degranulate alone but synergizes powerfully with every other activation receptor simultaneously, making the mast cell dramatically more sensitive to all other triggers. Elevated stem cell factor in chronically inflamed tissue may explain why reactions occur at exposures that would be tolerated in a less inflamed patient.
TLR4: The Gut Permeability Pathway Bacterial LPS translocating through a compromised gut barrier directly activates TLR4 on lamina propria mast cells. A direct mechanistic link between leaky gut and mast cell hyperreactivity that bypasses every conventional allergy pathway entirely.
A2B Receptor: The Hypoxia Amplifier Adenosine generated from ATP breakdown in hypoxic matrix tissue activates A2B receptors on mast cells, amplifying degranulation and cytokine secretion. Hypoxia in the matrix drives mast cell reactivity through multiple simultaneous mechanisms.
P2RX7: The Cellular Damage Pathway Extracellular ATP released from damaged cells in a poorly draining, congested matrix activates P2RX7 and triggers degranulation and IL-1β release.
The One Inhibitory Pathway
FcγRIIb engages allergen-specific IgG and suppresses IgE-driven mast cell activation. This is the mechanism behind allergen immunotherapy. When this pathway is functioning well it applies a meaningful brake on IgE reactivity.
What Every Non-IgE Pathway Has In Common
Every non-IgE activation pathway maps directly to degraded matrix conditions.
Acid activates ASIC and TRPV1, increasing Substance P and firing MRGPRX2.
Hypoxia directly triggers degranulation, activates TRPV1, upregulates FcγRI through IFN-γ, and generates adenosine that fires A2B receptors. Matrix damage and immune complex deposition activate complement generating C3a and C5a. Gut permeability releases LPS that fires TLR4. Cellular damage in poorly drained tissue releases ATP that fires P2RX7. Chronic inflammation elevates IFN-γ upregulating FcγRI and stem cell factor lowering the KIT threshold for every other receptor simultaneously.
The matrix is not simply where mast cells live. It is the source of every chronic activation signal they receive.
The Treatment Gap
Antihistamines block histamine after it is released from one pathway. Cromolyn and ketotifen prevent degranulation upstream of that primarily through the IgE calcium mechanism. Quercetin directly inhibits MRGPRX2 and raises the Substance P activation threshold, the most mechanistically precise natural intervention for the non-IgE pathway in the current literature.
None of these address the matrix soil that is generating the activation signals feeding every pathway simultaneously.
The Matrix Is the Soil Mast Cells Grow from
Every mast cell in the body lives directly in the extracellular matrix, the fluid-rich space between cells that fills connective tissue throughout every organ and system. This is not passive structural filler. The matrix is the biological soil that mast cells grow from, are fed by, and are continuously shaped by.
Just as a plant cannot thrive in depleted, toxic soil regardless of how much fertilizer it receives, a mast cell cannot calm down in a congested, acidic, hypoxic matrix regardless of how many antihistamines or stabilizers are prescribed. The soil has to change.
In the Matrix Medicine model we evaluate and restore five fundamental properties of matrix soil health:
pH balance - an alkaline matrix supports enzyme function, receptor sensitivity, and cellular repair. An acidic matrix activates sensory nerve acid receptors, increases Substance P production, and directly primes mast cells for reactivity.
Mineral status - magnesium, calcium, and trace minerals maintain the charge environment of the proteoglycan network that governs ion exchange and cellular signaling throughout the interstitium. Mineral depletion destabilizes matrix charge and impairs every cellular communication pathway.
Hydration - the matrix is a hydration-dependent structure. Dehydration thickens interstitial fluid, slows lymphatic flow, and reduces the conductivity through which every signal and nutrient must travel.
Oxygenation - oxygen tension in the matrix directly governs mitochondrial output and tissue regenerative capacity. Local hypoxia, which develops in congested, fibrotic matrix tissue, is a direct mast cell degranulation trigger and activates multiple additional inflammatory pathways simultaneously.
Drainage - the continuous lymphatic clearance of metabolic waste, inflammatory proteins, complement fragments, and cellular debris from the interstitium is the most foundational soil health property of all. When drainage stalls, every other property deteriorates. Waste accumulates. pH drops. Oxygen delivery fails. Complement fragments accumulate. The mast cell activation signals compound.
The Matrix Medicine Approach
Supporting the drainage pipeline is the foundational first step. The drainage pipeline is the network of organs responsible for clearing the matrix space- the lymphatic system, liver, kidneys, and gut. When this pipeline flows freely, the interstitium clears. When it stagnates, the matrix becomes the congested, acidic, hypoxic soil that keeps every non-IgE mast cell pathway continuously activated.
When we support the drainage pipeline:
Metabolic waste clears from the interstitium, removing the acid burden that activates sensory nerves and drives Substance P production. Complement-activating debris is removed from the matrix, reducing the continuous low-grade C3a and C5a generation firing mast cells through C3aR and C5aR.
LPS burden decreases as gut barrier integrity is restored, reducing TLR4 activation in the lamina propria. Extracellular ATP and adenosine accumulation reduces as cellular damage burden decreases, quieting P2RX7 and A2B receptor amplification. Matrix pH normalizes as acid metabolites clear, restoring the alkaline environment that calms sensory nerve firing.
Oxygenation improves as congestion resolves, removing the hypoxic trigger for direct degranulation and A2B amplification. The sensory nerve stops firing continuously. Substance P levels drop. The MRGPRX2 receptor stops being persistently stimulated. The mast cell returns to its homeostatic function - not because it was blocked or suppressed, but because the soil it lives in no longer signals threat.
This is not a replacement for medical management of MCAS. It is the foundational terrain layer beneath it that determines whether management produces resolution or only symptom suppression. The mast cell is not broken. The soil it lives in is. Restore the soil. The mast cell responds accordingly.
If you are a practitioner working with complex mast cell and chronic inflammatory patients and want to understand how to apply Matrix Medicine and drainage sequencing clinically, access my free Matrix Reset Masterclass here: https://courses.drmarkiwanicki.com/the-matrix-reset-masterclass.
If you are dealing with a chronic inflammatory condition and want to work on optimizing your matrix health personally, I work with patients one on one online to assess and restore the terrain driving chronic illness. You can learn more about working with me directly at https://drmarkiwanicki.com/work-with-me
References
Gilfillan AM, Austin SJ, Metcalfe DD. Mast cell biology: introduction and overview. Advances in Experimental Medicine and Biology. 2011;716:2-12. PMID 12217397
Woolhiser MR, Brockow K, Metcalfe DD. Activation of human mast cells by aggregated IgG through FcγRI: additive effects of C3a. Clinical Immunology. 2004;110(2):172-180. PMID 15003814
Tkaczyk C, Metcalfe DD, Gilfillan AM. Fcγ receptors on mast cells: activatory and inhibitory regulation of mediator release. International Archives of Allergy and Immunology. 2004;133(3):305-315. PMID 15017113
Azimi E, Reddy VB, Lerner EA. Brief communication: MRGPRX2, substance P, and the influence of mast cells on early and late phases of neurogenic inflammation. Journal of Investigative Dermatology. 2017;137(4):1638-1641. PMID 30686732
Ertas B, Ozyurt K, Atasoy M, Ustun H, Erikci A. MRGPRX2+ mast cells preferentially reside in proximity to and migrate toward substance P-positive nerve fibers. Frontiers in Immunology. 2023;14:1268991. PMC11492034
Binshtok AM, Wang H, Zimmermann K, et al. Nociceptors are intracellular targets of topical anesthetics. Journal of Neuroscience. 2007;27(39):10608-10612. PMC6790391
Soucek L, Lawlor ER, Soto D, Shchors K, Swigart LB, Evan GI. Mast cells are required for angiogenesis and macroscopic expansion of Myc-induced pancreatic islet tumors. Nature Medicine. 2007;13(10):1211-1218. PMID 12391033
Woolhiser MR, Brockow K, Metcalfe DD. C3a causes robust degranulation in human mast cells and stimulates chemokine MCP-1/CCL2 and RANTES/CCL5 production. Molecular Immunology. 2004;41(12):1235-1248. PMID 15607817
Gervais FG, Morello JP, Beaulieu M, et al. Regulation of human mast cell and basophil function by anaphylatoxins C3a and C5a. British Journal of Pharmacology. 2010;161(6):1217-1228. PMID 19895849
Nilsson G, Johnell M, Hammer CH, et al. C3a and C5a are chemotaxins for human mast cells and act through distinct receptors via a pertussis toxin-sensitive signal transduction pathway. Journal of Immunology. 1996;157(4):1693-1698. PMID 8759757
Gilfillan AM, Tkaczyk C. Integrated signalling pathways for mast-cell activation. Nature Reviews Immunology. 2006;6(3):218-230. PMC3395887
Avila G, Gonzalez-Espinosa C. Toll-like receptors and mast cells: mast cell activation and inflammatory responses to pathogens. Signal Transduction and Chemotaxis in Mast Cells. 2015;6:134. PMC4630209
Ryzhov S, Goldstein AE, Matafonov A, Zeng D, Biaggioni I, Feoktistov I. Adenosine-activated mast cells induce IgE synthesis by B lymphocytes. Frontiers in Immunology. 2021;12:670302. PMC8156042
Nagata N, Bhatt DL, Bhatt DL. Degranulation of mast cells as a target for drug development. International Journal of Molecular Sciences. 2023;24(10):8740. PMC10253146
Weng Z, Zhang B, Asadi S, et al. Quercetin is more effective than cromolyn in blocking human mast cell cytokine release and inhibits contact dermatitis and photosensitivity in humans. PLoS ONE. 2012;7(3):e33805. PMC3314669
Azimi E, Reddy VB, Lerner EA. Quercetin inhibits MRGPRX2-induced pseudo-allergic reaction via PLCγ-IP3R pathway. Journal of Dermatological Science. 2019;93(1):58-60. PMID 30471617
Zhu Y, Michalovich D, Wu H, et al. Mast cell stabilizers: from pathogenic roles to targeting therapies. Frontiers in Immunology. 2024;15:1418897. PMC11324444